This article reviews the current evidence and provides a practical, step-by-step approach for clinicians managing canine insulinoma cases. By the end of this article, readers should be able to:
- Describe the pathophysiology of insulinomas in dogs, including the mechanisms underlying hypoglycaemia.
- Identify the common signalment and clinical signs associated with insulinomas, including both acute and chronic presentations.
- Understand the diagnostic approach to canine insulinomas, including the interpretation of blood results the utility and limitations of different imaging modalities.
- Recognise the principles of tumour staging based on the WHO TNM classification and its relevance to prognosis.
- Outline the treatment options for canine insulinomas, including surgical and medical management, and describe potential perioperative complications.
- Evaluate the role of medical management in cases where surgery is not feasible or as an adjunct to surgical therapy.
Incidence and risk factors of canine insulinoma
Insulinomas arise from pancreatic β-cells and represent the most common pancreatic neoplasm in dogs (Tobin et al., 1999, Buishand, 2022). These tumours are frequently functional, secreting insulin in an unregulated manner, which leads to hyperinsulinaemia-induced hypoglycaemia (Kraai et al., 2025). In affected dogs, insulin secretion is not appropriately suppressed despite low blood glucose concentrations, due to a disruption of the normal negative feedback mechanisms in neoplastic β-cells. As a result, the hallmark of insulinoma is an inappropriately normal or elevated serum insulin concentration in the presence of hypoglycaemia (Buishand, 2022).
Dogs diagnosed with insulinomas have a mean age of 9.1 years, with reported cases ranging from 3 to 15 years of age (Mehlhaff et al., 1985, Tobin et al., 1999, Buishand, 2022). While there is no confirmed sex or breed predisposition, dogs of medium to large size appear to be overrepresented. Among these, certain breeds have been more commonly reported in published cases, such as German Shepherds, Irish Setters, Boxers, Golden Retrievers, Poodles, Fox Terriers, Collies, and Labrador Retrievers (Buishand, 2022).
Clinical signs of canine insulinoma
Although the clinical manifestations of insulinoma in dogs are primarily attributed to hypoglycaemia, immunohistochemical studies have shown that neoplastic β-cells may also produce a range of additional hormones, including glucagon, somatostatin, pancreatic polypeptide, growth hormone, insulin-like growth factor 1, gastrin, and serotonin (Hawkins et al., 1987, O'Brien et al., 1987). Nevertheless, mixed clinical syndromes involving these hormones have not been reported in dogs with endocrine pancreatic tumours (Buishand, 2022).
Neuroglycopenia refers to the effects of hypoglycaemia on the nervous system and accounts for the most common clinical manifestations of canine insulinomas. Affected dogs may present with weakness, ataxia, disorientation, behavioural changes, collapse or seizures. In addition, catecholamine release triggered by hypoglycaemia may lead to muscle tremors, shaking, anxiety and hunger. Clinical signs are often intermittent or episodic and may be precipitated by fasting, exercise, excitement or feeding. In chronic cases, clinical signs may be less pronounced despite markedly low blood glucose concentrations, and some dogs may appear clinically normal. Rarely, paraneoplastic peripheral neuropathy and brain lesions secondary to prolonged hypoglycaemia have been described (Shahar et al., 1985, Shimada et al., 2000). If hypoglycaemia is severe and sustained, cerebral cortical laminar necrosis may occur, potentially progressing to coma and death (Buishand, 2022).
Staging canine insulinoma
In dogs, insulinomas are considered malignant in the vast majority of cases (Leifer et al., 1986) and histological classification into adenoma or adenocarcinoma does not reliably predict biological behaviour (Minkus et al., 1997). In addition, at the time of diagnosis or surgical exploration, metastases are observed in approximately 40–50% of cases, most frequently involving regional lymph nodes and the liver; pulmonary metastases are rarely reported (Mehlhaff et al., 1985, Leifer et al., 1986). Primary insulinomas typically present as well-defined nodules smaller than 2.5 cm in diameter and are most commonly located in the left or right limb of the pancreas rather than in the pancreatic body (Mehlhaff et al., 1985).
Tumour staging of canine insulinomas follows the World Health Organization’s tumour-node-metastasis (TNM) classification system. This system categorises tumours based on the extent of the primary lesion, lymph node involvement, and presence of distant metastasis (Owen, 1980). A summary of the staging criteria is presented in Table 1.
Table 1. Staging of Canine Insulinomas According to the WHO TNM System (Owen 1980)
|
Stage |
Tumour (T) |
Lymph Nodes (N) |
Metastasis (M) |
Description |
|
Stage I |
T1 – Tumour confined to the pancreas |
N0 – No regional lymph node involvement |
M0 – No distant metastasis |
Tumour is limited to the pancreas, without spreading to lymph nodes or distant sites. |
|
Stage II |
T1 |
N1 – Regional lymph node involvement |
M0 |
Tumour has spread to regional lymph nodes, but there are no distant metastases. |
|
Stage III |
T0 (undetectable primary tumour) or T1 |
N0 or N1 |
M1 – Distant metastasis |
Tumour has metastasised to distant sites (e.g., liver or other organs). |
Diagnosis of insulinoma
A diagnosis of insulinoma is suspected in the presence of hypoglycaemia in conjunction with a normal or inappropriately elevated serum insulin concentration. If clinical suspicion for insulinoma exists but the dog is not hypoglycaemic at presentation, potentially due to stress-induced catecholamine release during hospitalisation, a monitored fasting protocol with serial blood glucose measurements every 30 to 60 minutes may be necessary to document this abnormality (Siliart and Stambouli, 1996, Buishand, 2022). Fasting should be carefully managed, as blood glucose levels typically decline before hypoglycaemic signs develop, potentially resulting in rapid and severe consequences in dogs with insulinoma. The majority of dogs with insulinomas will exhibit hypoglycaemia within 24 hours of controlled fasting (Buishand, 2022). Serum fructosamine concentration can also be measured in these cases, and values are expected to be lower than normal in dogs with chronic hypoglycaemia, which may support a suspicion of insulinoma, although this finding is not pathognomonic (Thoresen et al., 1995, Mellanby and Herrtage, 2002).
Imaging is frequently used to exclude other differential diagnosis of hypoglycaemia and for staging purposes. Thoracic and abdominal radiographs are typically unremarkable (Goutal et al., 2012) and abdominal ultrasonography, while commonly used, has limited diagnostic utility, successfully identifying pancreatic masses in fewer than 50% of cases and demonstrating low sensitivity and specificity for detecting metastases (Lamb et al., 1995, Tobin et al., 1999).
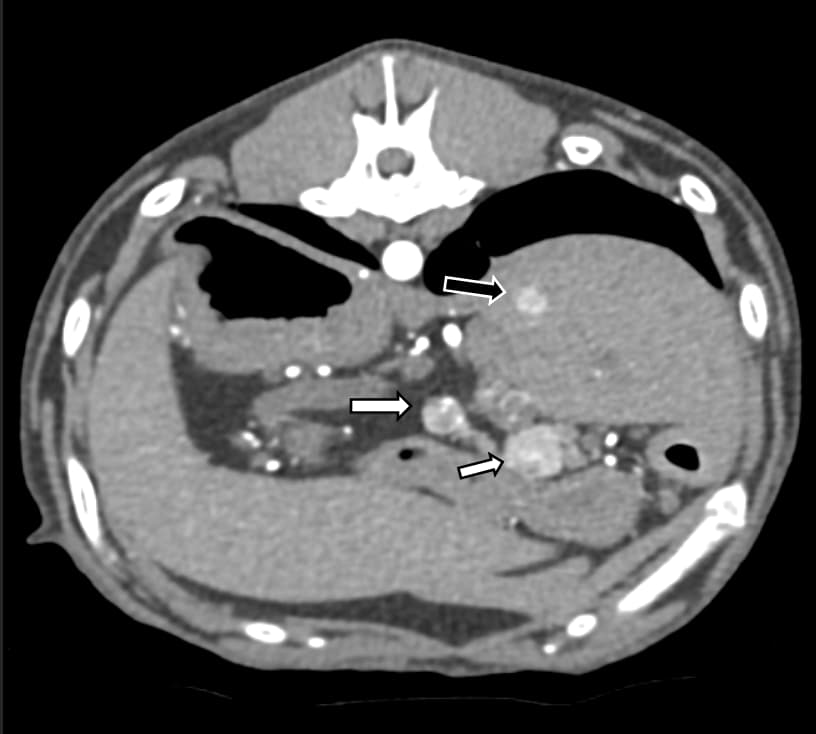
Computed tomography (CT) has been shown to be more sensitive than ultrasonography in detecting primary insulinomas in dogs, identifying 10 out of 14 confirmed lesions in one study (Robben et al., 2005). However, conventional pre- and post-contrast CT has limited sensitivity for metastatic disease and may yield false-positive results. More recently, contrast-enhanced CT (CECT) using dual- or triple-phase protocols, capturing arterial, portal and delayed venous phases, has improved the detection of insulinomas (Iseri et al., 2007, Buishand, 2022) (Figures 1 and 2). In human medicine, positron emission tomography is employed to localise insulinomas when standard imaging is inconclusive; however, its application in veterinary patients has yet to be evaluated (Sundin et al., 2007).

Figure 1. Post-contrast CT image of the abdomen of a dog, revealing two well-defined, contrast-enhancing nodules in the region of the pancreas (white arrows). These findings are consistent with pancreatic insulinomas. A small contrast-enhancing nodule in the liver (black arrow) is consistent with metastatic spread from the insulinomas.
.jpg?width=832&height=768&name=image%20(1).jpg)
Figure 2. Arterial-phase CT image of the abdomen of the same dog, demonstrating two large, contrast-enhancing nodules distributed throughout the hepatic parenchyma (white arrows), consistent with hepatic metastases secondary to pancreatic insulinomas.
Treatment options
Surgery
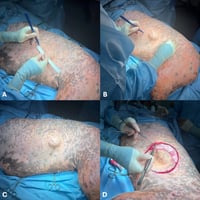
Surgical excision remains the treatment of choice for insulinomas when feasible (Leifer et al., 1986, Polton et al., 2007, Caywood et al., 1988, Tobin et al., 1999). Due to the limitations of current imaging modalities, exploratory laparotomy is often necessary to enable direct evaluation of the pancreas and a thorough inspection of the abdominal cavity to identify both the primary tumour and any metastatic lesions. Insulinomas are typically either visible or palpable during surgery and they occur with similar frequency in both pancreatic lobes (Nelson, 2015) (Figure 3). The surgical procedure usually involves a partial pancreatectomy (Figure 4), removing the tumour along with a margin of grossly normal tissue, using intracapsular dissection and vessel ligation or sealing techniques (Wouters et al., 2011). If metastases are present, debulking surgery may also be performed to reduce the tumour burden and improve glycaemic control (Buishand, 2022).
.jpg?width=839&height=926&name=image%20(2).jpg)
Figure 3: Intra-operative picture of an insulinoma (arrow) adjacent to the left limb of the pancreas (star)
.jpg?width=888&height=1008&name=image%20(3).jpg)
Figure 4: Partial left pancreatectomy
Postoperative complications can include persistent hypoglycaemia, acute pancreatitis (which occurs in approximately 10% of cases) (Del Busto et al., 2020), and, less frequently, hyperglycaemia (Nelson, 2015, Buishand, 2022). While overt pancreatitis is rare, inappetence and vomiting were noted in 27.3% and 24.2% of dogs, respectively, in a study of 33 surgical cases (Hixon et al., 2019). Persistent hypoglycaemia is observed in up to 23% of dogs, often indicating the presence of residual tumour tissue, which typically carries a poor prognosis (Del Busto et al., 2020). Postoperative hyperglycaemia is usually temporary, though a small number of patients may require insulin therapy (Nelson, 2015).
Postoperative care should include intravenous fluid therapy, multimodal analgesia, antiemetics and vigilant monitoring of blood glucose concentrations, as well as clinical signs of pancreatitis. Additionally, all excised or biopsied tissues should be submitted for histopathological evaluation to confirm the diagnosis and guide further management (Goutal et al., 2012).
Medical Management
Medical management of this neoplasm may be used prior to surgery, as an adjunct to surgical treatment or when surgery is not feasible.
Perioperative in-hospital management
Several important factors must be considered in the perioperative management of dogs with insulinoma. Preoperative stabilisation is essential and often involves treating acute hypoglycaemia (Smith and Harkin, 2000, Datte et al., 2016). This is typically managed with intravenous dextrose, initially administered as a slow bolus (1 ml/kg of 20% glucose over 5 to 10 minutes), followed by a continuous rate infusion (2.5 to 5% glucose at 3–4 mL/kg/h) (Buishand, 2022). However, this approach may stimulate further unregulated insulin secretion, potentially exacerbating hypoglycaemia. In cases of hyperinsulinemic-hypoglycemic crisis, glucagon infusions (50 ng/kg bolus, followed by a continuous infusion of 5–10 ng/kg/min) have been used safely and effectively in some patients (Smith and Harkin, 2000, Datte et al., 2016, Buishand, 2022). Additionally, extended periods of fasting should be avoided to prevent further worsening of hypoglycaemia (Goutal et al., 2012).
Outpatient medical management
One commonly used option is prednisone, which has insulin-antagonistic, gluconeogenic and glycogenolytic properties. Treatment typically begins at a dose of 0.25 mg/kg orally twice daily, with adjustments made based on the patient’s response. If prednisone alone is insufficient, diazoxide can be added. This benzothiadiazine derivative inhibits insulin release and promotes hepatic gluconeogenesis and glycogenolysis. It is usually started at 5 mg/kg orally twice daily and can be titrated up to a total daily dose of 30 mg/kg. Around 70% of dogs respond to diazoxide, although side effects such as vomiting, ptyalism, anorexia and diarrhoea may occur. Another therapeutic option is octreotide, a somatostatin analogue that suppresses insulin secretion by pancreatic beta cells. It has been effective in approximately 50% of dogs with insulinoma. The typical dose ranges from 10 to 50 μg subcutaneously, administered two to three times daily. Side effects appear to be rare and recent studies have shown that octreotide can significantly reduce plasma insulin levels and increase blood glucose concentrations. However, further research is needed to fully understand its long-term efficacy (Nelson, 2015, Robben et al., 2006).
Chemotherapy
Streptozocin, though infrequently used, is the most commonly reported chemotherapeutic agent in dogs. Historically limited by its nephrotoxic potential (Meyer, 1976), more recent studies indicate that co-administration with aggressive saline diuresis significantly reduces this risk (Moore et al., 2002). Adverse effects may include vomiting, hyperglycemia, diabetes mellitus, elevated liver enzymes, and mild hematologic abnormalities (Northrup et al., 2013). While streptozocin has shown occasional benefits such as tumour reduction or improvement in paraneoplastic neuropathy, it has not demonstrated a significant extension in normoglycemia duration compared to other medical or surgical approaches (Moore et al., 2002). Given the relative high risk of possible side effects, its overall risk–benefit ratio remains uncertain (Nelson, 2015). More recently, a case report and two small retrospective case series have described the use of the tyrosine kinase inhibitor toceranib phosphate (Palladia®) in dogs with insulinoma (Flesner et al., 2019, Alonso-Miguel et al., 2021, Sheppard-Olivares et al., 2022). While some patients achieved long-term glycaemic control during treatment, it remains uncertain whether this response was directly attributable to the drug (Buishand, 2022). The authors typically recommend treatment with toceranib in cases where surgical excision is not feasible or when the disease progresses, either alone or in combination with corticosteroids.
Dietary management for canine insulinoma
Dietary management plays a key role in the care of dogs with insulinoma. A diet of small, frequent meals rich in fat, protein and complex carbohydrates is recommended, while simple sugars should be avoided (Nelson, 2015). In addition, exercise should be limited to short, controlled leash walks to reduce the risk of hypoglycaemia. If clinical signs persist despite these dietary and physical activity modifications, further medical therapy may be necessary (Buishand, 2022).
A summary of the medical treatment options, including doses and potential side effects, is provided in Table 2 below.
Table 2. Medical Treatment Options for Canine Insulinoma
|
Category |
Drug |
Dose |
Route |
Frequency |
Notes |
|
Perioperative management |
Dextrose |
1 mL/kg of 20% over 5–10 min, then 2.5–5% at 3–4 mL/kg/h |
IV |
Bolus + CRI |
May stimulate unregulated insulin secretion and exacerbate hypoglycaemia |
|
|
Glucagon |
50 ng/kg bolus, then 5–10 ng/kg/min |
IV |
Bolus + CRI |
Used in hyperinsulinemic-hypoglycaemic crisis; safe and effective in some patients |
|
|
Prednisone |
0.25 mg/kg |
PO |
q12h |
Insulin-antagonistic, gluconeogenic, and glycogenolytic properties
|
|
Outpatient management |
Prednisone |
0.25 mg/kg |
PO |
q12h |
Insulin-antagonistic, gluconeogenic, and glycogenolytic properties |
|
|
Diazoxide |
5–30 mg/kg |
PO |
q12h |
Inhibits insulin release; promotes gluconeogenesis and glycogenolysis; side effects: GI signs |
|
|
Octreotide |
10–50 μg |
SC |
q12h to q8h |
Suppresses insulin secretion; effective in ~50% of dogs; side effects are rare |
|
Chemotherapy |
Streptozocin |
500 mg/m |
IV |
Every 3 weeks |
Risk of nephrotoxicity; should be combined with a protocol for induction of diuresis; unclear efficacy |
|
|
Toceranib phosphate |
2.1–3.27 mg/kg |
PO |
MWF or EOD |
Median starting dose intensity: 8.1 mg/kg/week (min–max: 6.30–11.38) |
IV = intravenous; PO = oral; SC = subcutaneous; CRI = constant rate infusion; q12h = every 12 hours; q8h = every 8 hours; MWF = Monday, Wednesday, Friday; EOD = every other day; GI = gastrointestinal. Doses and treatment protocols adapted from: Buishand (2022), Datte et al. (2016), Smith & Harkin (2000), Goutal et al. (2012), Nelson (2015), Robben et al. (2006), Meyer (1976), Moore et al. (2002), Northrup et al. (2013), Alonso-Miguel et al. (2021), Flesner et al. (2019), Sheppard-Olivares et al. (2022).
Canine insulinoma prognosis
The short-term prognosis for dogs with insulinoma is generally favourable; however, the long-term outlook remains guarded due to the risk of metastasis or tumour regrowth. Dogs treated with surgery followed by medical management are more likely to achieve and maintain euglycemia and demonstrate prolonged survival compared to those managed medically alone (Tobin et al., 1999, Polton et al., 2007, Buishand, 2022). Reported median survival times (MSTs) following partial pancreatectomy typically range from 12 to 14 months (Schoeman, 2017). Additionally, prognosis is closely associated with clinical staging (Nelson, 2015), with stage I disease conferring a longer disease-free interval (DFI) than stages II or III. Approximately 50% of dogs with stage I disease remain euglycemic at 14 months postoperatively, compared to fewer than 20% of those with more advanced disease.
On the other hand, dogs with stage III disease exhibit significantly shorter survival, with around half succumbing within 6 months (Caywood et al., 1988). More recent retrospective data suggests improved outcomes, reporting a median DFI of 496 days and MST of 785 days in a cohort of 19 dogs undergoing surgical resection, 9 of which received adjunctive medical treatment following partial pancreatectomy. This subgroup received medical therapy after postsurgical relapse, indicated by the recurrence of clinical signs or laboratory-confirmed hypoglycaemia, and had an MST of 1316 days. In contrast, dogs treated with medical therapy alone had a considerably shorter MST of 196 days. However, the overall MST for all dogs receiving medical therapy, regardless of whether it was administered alone or following surgery, was 452 days, measured from the time medical therapy was initiated (Polton and others 2007). These findings underscore the value of combining surgical and medical approaches, particularly in cases with recurrence of clinical signs.
Furthermore, the Ki67 proliferation index has emerged as a potential prognostic biomarker in canine insulinomas. A Ki67 index > 2.5% is associated with significantly shorter disease-free intervals (DFIs) and survival times (STs) compared to tumours with an index ≤ 2.5% (Buishand et al., 2010). Dogs that are normoglycaemic or develop postoperative hyperglycaemia tend to have significantly longer median survival times (680 days) compared to those that remain hypoglycaemic after surgery (90 days), highlighting the prognostic value of postoperative glycaemic status (Trifonidou et al., 1998).
References (click to expand)
ALONSO-MIGUEL, D., GARCÍA-SAN JOSÉ, P., GONZÁLEZ SANZ, S., CLARÉS MORAL, I. & PÉREZ-ALENZA, M. D. 2021. Evaluation of palliative therapy, alone or in combination with toceranib phosphate, in dogs diagnosed with metastatic or recurrent beta-cell neoplasia. N Z Vet J, 69, 234-239.
BUISHAND, F. O. 2022. Current Trends in Diagnosis, Treatment and Prognosis of Canine Insulinoma. Vet Sci, 9.
BUISHAND, F. O., KIK, M. & KIRPENSTEIJN, J. 2010. Evaluation of clinico-pathological criteria and the Ki67 index as prognostic indicators in canine insulinoma. Vet J, 185, 62-7.
CAYWOOD, D., KLAUSNER, J., O'LEARY, T., WITHROW, S., RICHARDSON, R., HARVEY, H., NORRIS, A., HENDERSON, R. & JOHNSTON, S. 1988. Pancreatic insulin-secreting neoplasms: clinical, diagnostic, and prognostic features in 73 dogs. Journal of the American Animal Hospital Association, 24, 577–584.
DATTE, K., GUILLAUMIN, J., BARRETT, S., MONNIG, A. & COOPER, E. 2016. Retrospective evaluation of the use of glucagon infusion as adjunctive therapy for hypoglycemia in dogs: 9 cases (2005-2014). J Vet Emerg Crit Care (San Antonio), 26, 775-781.
DEL BUSTO, I., GERMAN, A. J., TREGGIARI, E., ROMANELLI, G., O'CONNELL, E. M., BATCHELOR, D. J., SILVESTRINI, P. & MURTAGH, K. 2020. Incidence of postoperative complications and outcome of 48 dogs undergoing surgical management of insulinoma. J Vet Intern Med, 34, 1135-1143.
FLESNER, B. K., FLETCHER, J. M., SMITHEE, T. & BOUDREAUX, B. 2019. Long-Term Survival and Glycemic Control with Toceranib Phosphate and Prednisone for a Metastatic Canine Insulinoma. J Am Anim Hosp Assoc, 55, e55105.
GOUTAL, C. M., BRUGMANN, B. L. & RYAN, K. A. 2012. Insulinoma in dogs: a review. J Am Anim Hosp Assoc, 48, 151-63.
HAWKINS, K. L., SUMMERS, B. A., KUHAJDA, F. P. & SMITH, C. A. 1987. Immunocytochemistry of normal pancreatic islets and spontaneous islet cell tumors in dogs. Vet Pathol, 24, 170-9.
HIXON, L. P., GRIMES, J. A., WALLACE, M. L. & SCHMIEDT, C. W. 2019. Risk factors for gastrointestinal upset and evaluation of outcome following surgical resection of canine pancreatic β-cell tumors. Can Vet J, 60, 1312-1318.
ISERI, T., YAMADA, K., CHIJIWA, K., NISHIMURA, R., MATSUNAGA, S., FUJIWARA, R. & SASAKI, N. 2007. Dynamic computed tomography of the pancreas in normal dogs and in a dog with pancreatic insulinoma. Vet Radiol Ultrasound, 48, 328-31.
KRAAI, K., O'NEILL, D. G., DAVISON, L. J., BRODBELT, D. C., GALAC, S. & BUISHAND, F. O. 2025. Incidence and risk factors for insulinoma diagnosed in dogs under primary veterinary care in the UK. Sci Rep, 15, 2463.
LAMB, C. R., SIMPSON, K. W., BOSWOOD, A. & MATTHEWMAN, L. A. 1995. Ultrasonography of pancreatic neoplasia in the dog: a retrospective review of 16 cases. Vet Rec, 137, 65-8.
LEIFER, C. E., PETERSON, M. E. & MATUS, R. E. 1986. Insulin-secreting tumor: diagnosis and medical and surgical management in 55 dogs. J Am Vet Med Assoc, 188, 60-4.
MEHLHAFF, C. J., PETERSON, M. E., PATNAIK, A. K. & CARRILLO, J. M. 1985. Insulin-producing islet cell neoplasms: Surgical considerations and general management in 35 dogs. J. Am. Anim. Hosp. Assoc., 21, 607–612.
MELLANBY, R. J. & HERRTAGE, M. E. 2002. Insulinoma in a normoglycaemic dog with low serum fructosamine. J Small Anim Pract, 43, 506-8.
MEYER, D. J. 1976. Pancreatic islet cell carcinoma in a dog treated with streptozotocin. Am J Vet Res, 37, 1221-3.
MINKUS, G., JÜTTING, U., AUBELE, M., RODENACKER, K., GAIS, P., BREUER, W. & HERMANNS, W. 1997. Canine neuroendocrine tumors of the pancreas: a study using image analysis techniques for the discrimination of metastatic versus nonmetastatic tumors. Vet Pathol, 34, 138-45.
MOORE, A. S., NELSON, R. W., HENRY, C. J., RASSNICK, K. M., KRISTAL, O., OGILVIE, G. K. & KINTZER, P. 2002. Streptozocin for treatment of pancreatic islet cell tumors in dogs: 17 cases (1989-1999). J Am Vet Med Assoc, 221, 811-8.
NELSON, R. W. 2015. Beta-cell neoplasia: insulinoma. Canine and Feline Endocrinology. 4th ed. St. Louis, MO: Elsevier.
NORTHRUP, N. C., RASSNICK, K. M., GIEGER, T. L., KOSAREK, C. E., MCFADDEN, C. W. & ROSENBERG, M. P. 2013. Prospective evaluation of biweekly streptozotocin in 19 dogs with insulinoma. J Vet Intern Med, 27, 483-90.
O'BRIEN, T. D., HAYDEN, D. W., O'LEARY, T. P., CAYWOOD, D. D. & JOHNSON, K. H. 1987. Canine pancreatic endocrine tumors: immunohistochemical analysis of hormone content and amyloid. Vet Pathol, 24, 308-14.
OWEN, L. N. 1980. T.N.M. Classification of Tumours in Domestic Animals, Geneva, Switzerland, World Health Organization.
POLTON, G. A., WHITE, R. N., BREARLEY, M. J. & EASTWOOD, J. M. 2007. Improved survival in a retrospective cohort of 28 dogs with insulinoma. J Small Anim Pract, 48, 151-6.
ROBBEN, J. H., POLLAK, Y. W., KIRPENSTEIJN, J., BOROFFKA, S. A., VAN DEN INGH, T. S., TESKE, E. & VOORHOUT, G. 2005. Comparison of ultrasonography, computed tomography, and single-photon emission computed tomography for the detection and localization of canine insulinoma. Journal of Veterinary Internal Medicine, 19, 15–22.
ROBBEN, J. H., VAN DEN BROM, W. E., MOL, J. A., VAN HAEFTEN, T. W. & RIJNBERK, A. 2006. Effect of octreotide on plasma concentrations of glucose, insulin, glucagon, growth hormone, and cortisol in healthy dogs and dogs with insulinoma. Res Vet Sci, 80, 25-32.
SCHOEMAN, J. 2017. Insulin-secreting tumors. In: ETTINGER SJ, F. E. (ed.) Textbook of Veterinary Internal Medicine. 8th ed. St. Louis: Elsevier.
SHAHAR, R., ROUSSEAUX, C. & STEISS, J. 1985. Peripheral polyneuropathy in a dog with functional islet B-cell tumor and widespread metastasis. J Am Vet Med Assoc, 187, 175-7.
SHEPPARD-OLIVARES, S., BELLO, N. M., JOHANNES, C. M., HOCKER, S. E., BILLER, B., HUSBANDS, B., SNYDER, E., MCMILLAN, M., MCKEE, T. & WOUDA, R. M. 2022. Toceranib phosphate in the management of canine insulinoma: A retrospective multicentre study of 30 cases (2009-2019). Vet Rec Open, 9, e27.
SHIMADA, A., MORITA, T., IKEDA, N., TORII, S. & HARUNA, A. 2000. Hypoglycaemic brain lesions in a dog with insulinoma. J Comp Pathol, 122, 67-71.
SILIART, B. & STAMBOULI, F. 1996. Laboratory diagnosis of insulinoma in the dog: a retrospective study and a new diagnostic procedure. J Small Anim Pract, 37, 367-70.
SMITH, S. A. & HARKIN, K. R. 2000. Glucagon constant-rate infusion: a novel strategy for the management of hyperinsulinemic-hypoglycemic crisis in the dog. Journal of the American Animal Hospital Association, 36, 27–32.
SUNDIN, A., GARSKE, U. & ORLEFORS, H. 2007. Nuclear imaging of neuroendocrine tumours. Best Pract Res Clin Endocrinol Metab, 21, 69-85.
THORESEN, S. I., ALEKSANDERSEN, M., LØNAAS, L., BREDAL, W. P., GRØNDALEN, J. & BERTHELSEN, K. 1995. Pancreatic insulin-secreting carcinoma in a dog: fructosamine for determining persistent hypoglycaemia. J Small Anim Pract, 36, 282-6.
TOBIN, R. L., NELSON, R. W., LUCROY, M. D., WOOLDRIDGE, J. D. & FELDMAN, E. C. 1999. Outcome of surgical versus medical treatment of dogs with beta cell neoplasia: 39 cases (1990-1997). J Am Vet Med Assoc, 215, 226-30.
TRIFONIDOU, M. A., KIRPENSTEIJN, J. & ROBBEN, J. H. 1998. A retrospective evaluation of 51 dogs with insulinoma. Vet Q, 20 Suppl 1, S114-5.
WOUTERS, E. G., BUISHAND, F. O., KIK, M. & KIRPENSTEIJN, J. 2011. Use of a bipolar vessel-sealing device in resection of canine insulinoma. J Small Anim Pract, 52, 139-45.